Tough targets. Confident decisions.
Powered by Spectral Shift technology
-
Dianthus
- Applications
- Technology
- System
- Software
- Consumables
Dianthus is a plate-based and microfluidics-free affinity screening platform. Measurements are in solution and mass-independent — ideal for succeeding at demanding screening campaigns.

It’s not surprising that the toughest drug targets are often the hardest to measure. The Dianthus™ system makes it possible with fast, in-solution binding data from real samples. With the Dianthus α applications package, it delivers deeper insight into protein stability and complex binding behaviors empowering scientists to explore targets once considered out of reach.
Finally find success with your challenging affinity screening campaigns
De-risk early development
Accelerate time to insight with a 12-point binding curve in one minute or screen 2,000 fragments in just three hours. Detect aggregation and folding issues early with in-solution data you can trust, even from fragile targets. With sample use as low as 10–20 μL per data point and detection limits down to 250 pM, conserve material while gaining high-quality results.
Validate hits with confidence
Some affinity screening methods only generate good data with pure samples, which delays projects and puts a lot of pressure on screening teams. With Dianthus you get high-quality data from real-life samples, so aggregates, impurities, or precipitates won’t hold you back.
Push the boundaries of what’s druggable
Go beyond traditional approaches with a binding site–agnostic platform. Characterise ternary complexes, study covalent modifications, and resolve slow binding events with measurable kinetics (kobs ≤ 1 × 10⁻² s⁻¹). Expand discovery to targets once considered undruggable.
Beat common concerns encountered with other technologies
No matter what screening campaign you’re tackling, you are bound to encounter common issues that worry most scientists. Relax and focus on your research knowing that with Dianthus, you don’t have to fret about them.
Measure the broadest range of affinities
Dianthus detects a wide range of binding affinities — picomolar to millimolar — so you catch very strong and weak binders.
Characterize in solution, no immobilization required
Analyzing interactions in close-to-native conditions is ideal, especially when dealing with challenging targets. Never worry about negatively impacting your target’s binding site or lacking control over equilibrium conditions, since measurements are done in solution.
Gain deeper insight with Dianthus α applications package
Optical Unfolding and Slow Kinetics capabilities reveal stability and complex interactions that conventional methods miss.
Consume small amounts of target and compounds
Every little bit counts. Saving on costly samples and library compounds means you can do more screening campaigns or projects.
Automate your affinity screening campaigns
Get hours of uninterrupted and unattended operation with a format that’s compatible with many automation solutions.
Take your analysis further with structured, AI-ready data exports
Human and machine readable .json file formats allow for rapid integration of instrument data into your analysis pipeline, whether that’s NanoTemper software, alternative vendors, or open source options.
Lean on Dianthus when your affinity screening campaigns encounter roadblocks
Don’t give up just yet when you’re faced with a complicated affinity screening campaign. Armed with Dianthus, you have an opportunity to keep your project moving forward when your hit identification or lead optimization involves any of these molecules.
PROTACs and other small molecule protein degraders
Development of protein degraders is a multi-step and complex process. Selecting the appropriate biochemical and biophysical methods for each step is crucial to move the drug candidates down the pipeline and to market faster and with confidence.
Dianthus resolves the challenges presented by the characterization of binary and ternary complexes and the determination of cooperativity.
Learn how to use Dianthus for PROTACs
Fragment libraries
Challenge
Because your method requires a significant change in mass to measure interactions, it’s difficult to identify hits from a library of fragments due to their very low molecular weight.
Solution
With Dianthus, you identify hits from fragment libraries with confidence because measurements are mass-independent.
And, because Dianthus detects a broad range of affinity strengths, it’s useful for hit identification when fragments bind to proteins with mM affinity and lead optimization when fragments grow to form larger compounds that bind at low nM affinities.
Intrinsically disordered proteins (IDPs) and other aggregation-prone targets
-
Challenge
Because IDPs don’t fold into a homogeneous 3D structure, you see aggregation triggered by non-native intermolecular interactions. On top of that, you are concerned with aggregation resulting from the high concentrations required by other methods. -
Solution
With Dianthus, a high concentration is not required like it is with ITC. And if aggregation happens at low concentration, Dianthus differentiates between binding and aggregation so you get a better understanding of how the molecules behave -
Challenge
Your method requires immobilization which easily disturbs the conformational equilibrium of IDPs. -
Solution
Dianthus measures in solution, so the conformational equilibrium is not at risk of being disrupted as it happens with methods that require immobilization like SPR.
Dianthus is your trusted screening platform from start to finish
Hit Identification
Finding true hits faster is the most important step in making your drug discovery workflow efficient. With Dianthus, you’ll find hits quickly and move on to hit validation confidently, whether it’s fragment-based or small molecule single-dose screening.
Lead Validation
Spend less time sorting through the strong and weak binders. Dianthus generates easy-to-interpret affinity ranking tables and histograms to help you quickly decide on the right candidates and start lead optimization sooner.
Lead Optimization
Once validation is complete, it’s time to improve target specificity, selectivity, and potency. Use Dianthus to verify that binding affinities remain strong. This, combined with your ADME, toxicity, and PK/PD results, ensures you’re developing the best drug candidates.
Validate hits with a platform that offers two biophysical technologies
Orthogonal validation with multiple biophysical methods is critical for every primary screening campaign. It’s common to find hits with one method that turn out to be aggregators when tested by another. That’s why orthogonal validation is key to building confidence in your results.
Dianthus delivers the clarity scientists need through two fluorescence-based technologies that now provide deeper insight into molecular interactions. It reveals stability changes, detects subtle unfolding or aggregation events, and captures complex binding behaviors that develop over time.
Ensure your success with these two biophysical modalities
Measuring challenging interactions between various types of molecules at times requires a different approach. So having two different modalities in the same instrument helps you cover all the types of interactions you encounter. Dianthus comes with two biophysical modalities — Spectral Shift and Temperature Related Intensity Change (TRIC) — that help you measure the strength of your challenging molecular interactions.
-
Spectral Shift
-
TRIC
Spectral Shift
Dianthus is the first affinity screening platform to use Spectral Shift. The notion of spectral shift isn’t new, but Dianthus is the first platform that uses it to derive affinity constants.
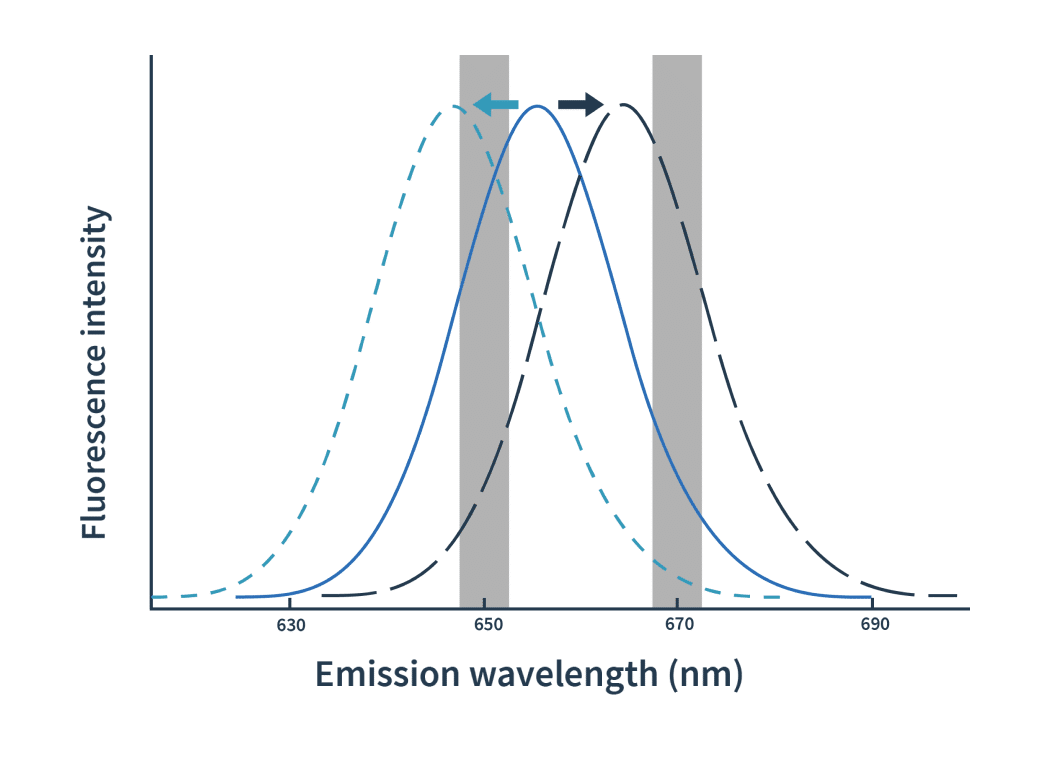
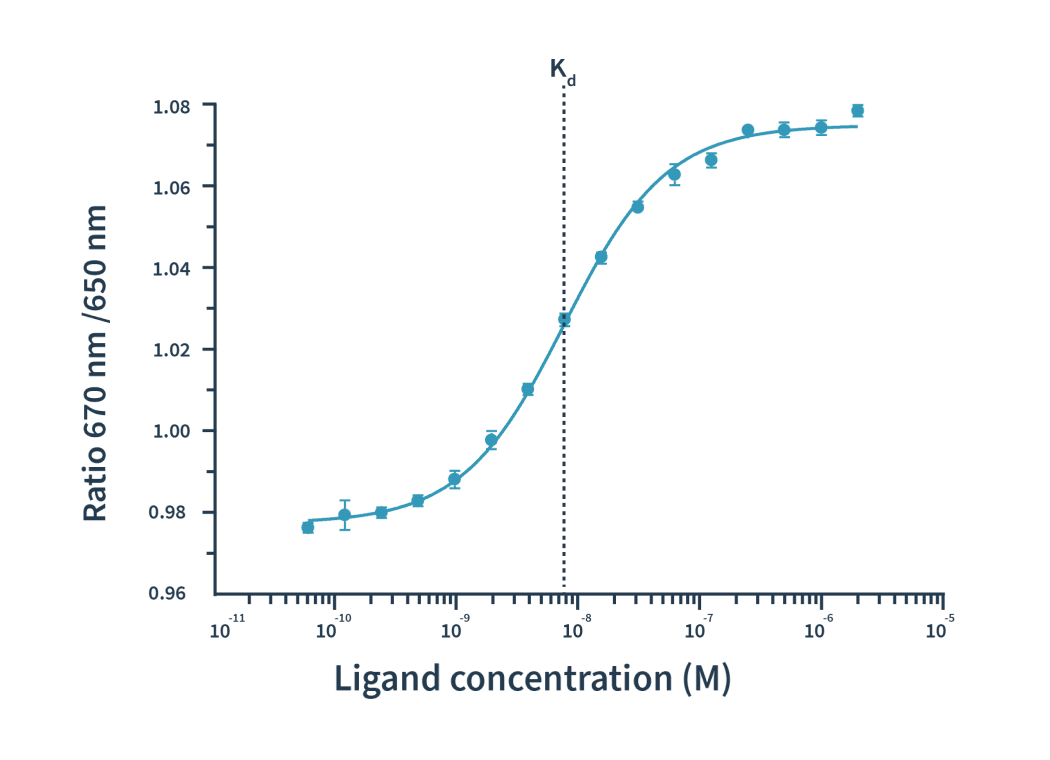
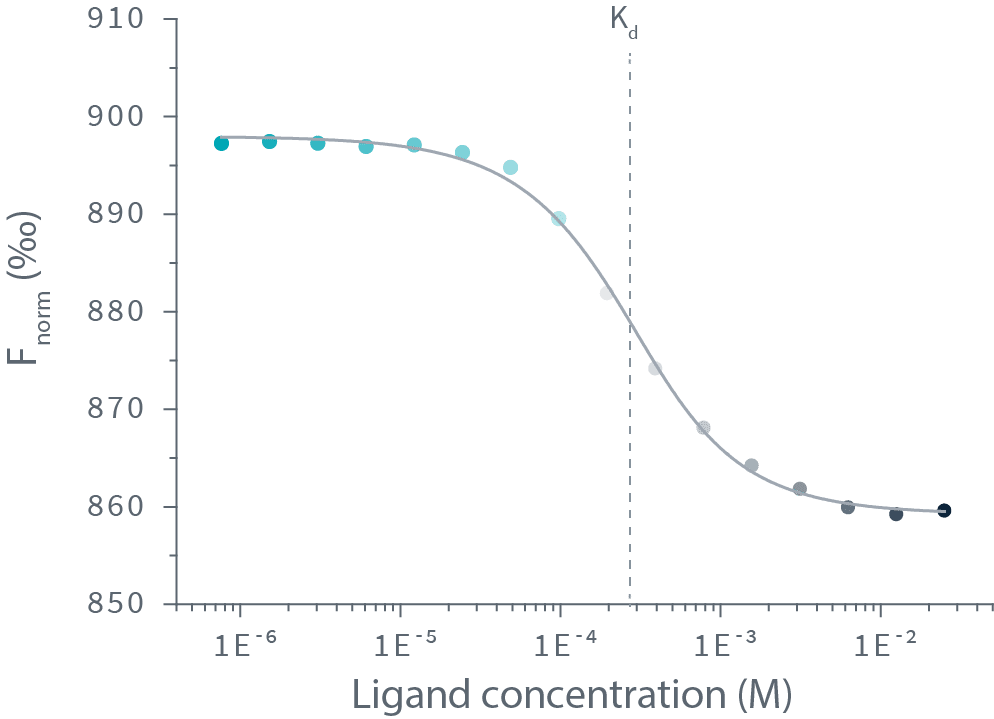
The process is actually pretty simple: label one binding partner with a fluorophore, then mix a fixed amount of it with a dilution series of the ligand. After excitation at 590nm of the mix, the binding events are detected as shifts of spectra towards either blue or red. The detection of spectral shifts is achieved through dual-wavelength detection at exactly 650nm and 670nm in an isothermal environment — this makes Dianthus so precise and able to detect very subtle shifts.
Then the Kd is calculated by plotting the ratios of the fluorescence intensities from the two wavelengths against the ligand concentration.
Optical Unfolding
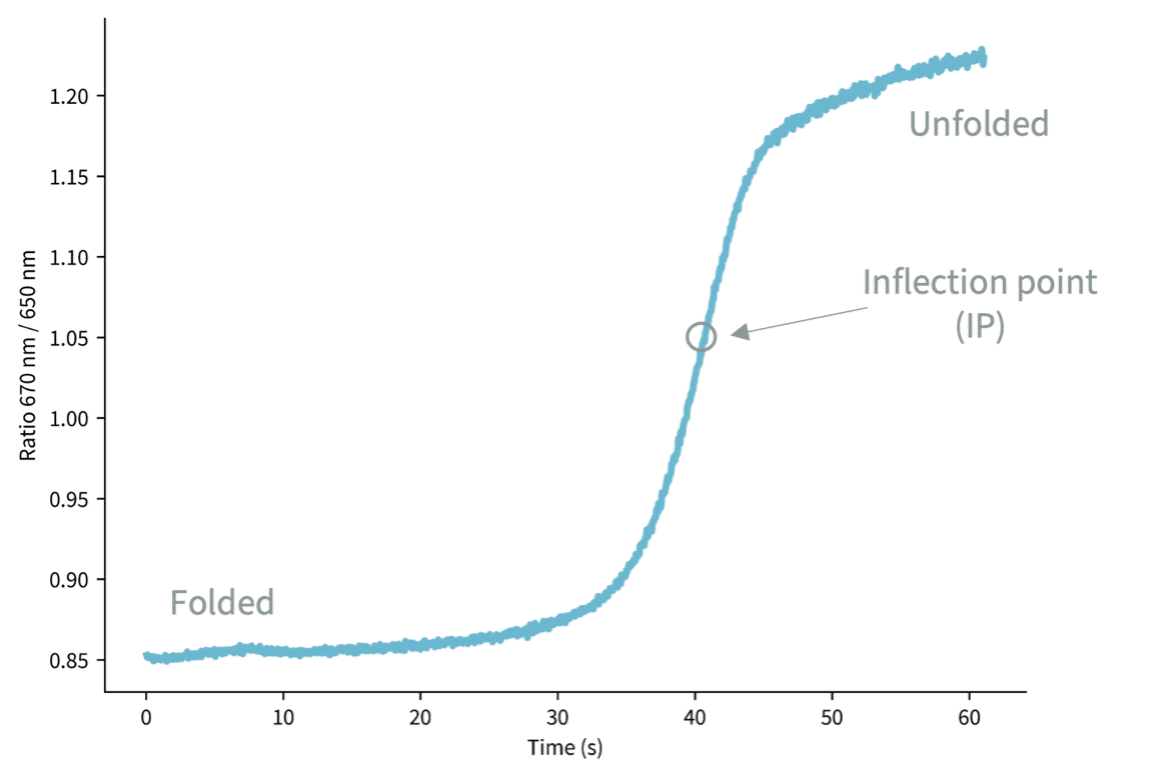
Dianthus also enables thermal shift assays, where inflection time correlates directly with the thermal stability of the target protein. By monitoring the unfolding profile, researchers can assess target stability during screening campaigns and detect changes upon ligand binding. When applied to selected hits from a primary screen, thermal shift assays serve as an orthogonal validation method, confirming true binding events and helping to rule out false positives. This makes Dianthus a powerful tool not only for affinity determination, but also for building confidence in screening results through complementary stability data.
TRIC
Use TRIC — a proven technology that’s been around for over 10 years that compliments Spectral Shift
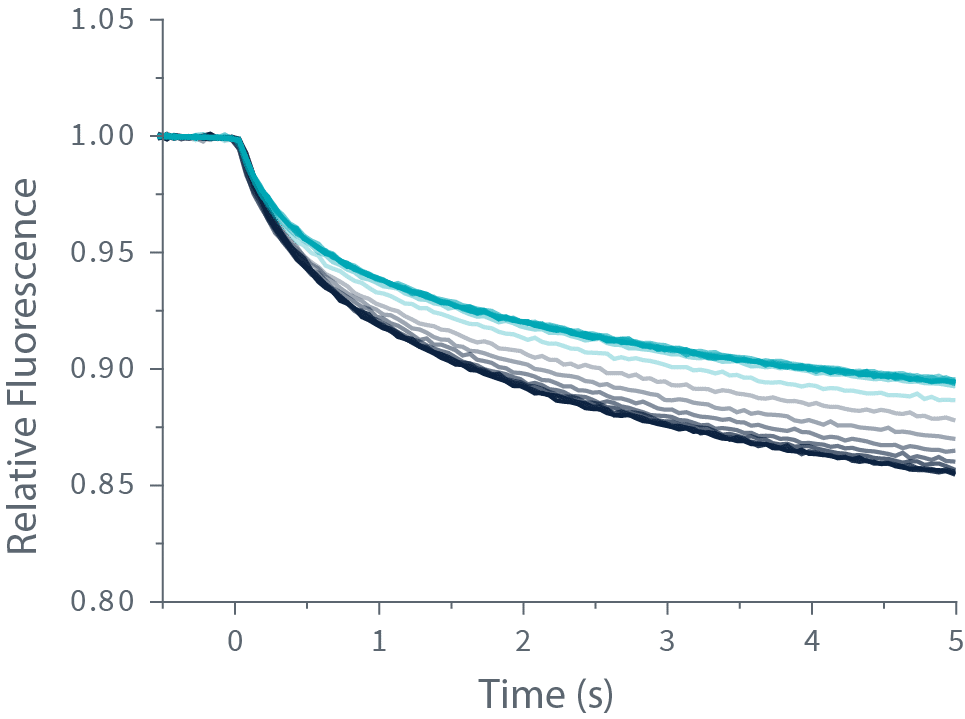
Quantifying molecular interactions with TRIC is done by labeling your target molecule with a fluorescent dye and mixing it with your ligand. Then, a very precise and brief laser-induced temperature change is applied, which amplifies the variation in fluorescence intensity caused by the ligand binding to your target.
The change in fluorescence is measured and plotted against the ligand concentration to obtain the dissociation constant or Kd.

Integrate Dianthus into your existing automation solution
Dianthus is a plate-based, microfluidics-free screening platform that easily integrates into any automated setup via gRPC framework. And with no regular maintenance, your projects don’t get delayed due to downtime. Dianthus is ready whenever you need it — non-stop, 24/7.
Decide which hits are worth moving forward with
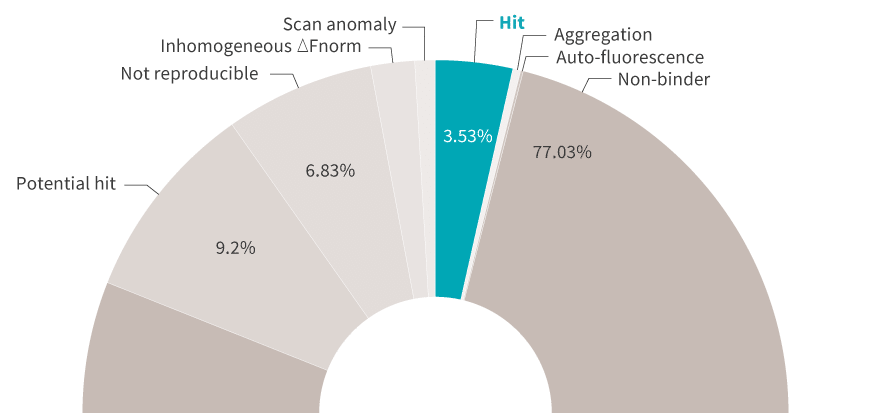
Generating results is great, but getting automated, actionable insights from your results is even better. DI.Screening Analysis software gives you screening summaries as well as easy-to-interpret ranking tables and histograms. And because your Kds are calculated from data with high signal-to-noise ratios, you decide with certainty which candidates are worth moving forward with.
Get consistent results with high-quality consumables

Get consistent results with high-quality consumables
Get the high quality and consistency you expect from consumables when finding and validating hits. Dianthus 384-well plates have all the features needed for the best performance and user experience:
- They are manufactured with a proprietary coating that prevents protein from sticking to the wells
- They go through rigorous QC testing to ensure consistency from well to well
- They have a barcode that helps you track your assay and data back to a specific plate.
You’ll also want to use a labeling kit to label your proteins or peptides with fluorophores selected for their sensitivity to binding events — so you get the best results with Spectral Shift or TRIC.
